Innovation
with social
and economic impact

Innovating for
the common good
Discover, through real solutions and everyday situations, how the technologies we develop at Eurecat enhance the competitiveness of businesses, the sustainability of the planet, and, above all, people’s lives.
We are the technological reference of companies for their innovation challenges. Talent, experience and infrastructures at the service of business competitiveness and environmental and social well-being.
companies
Catalonia
Madrid
and Málaga
in LATAM
are SMEs
and HE projects
Activities
We create and/or improve products, services and processes, from the idea to industrialization.
Knowledge areas
We integrate multi-technological and multidisciplinary capabilities to address complex business challenges in a comprehensive and sustainable way.

eurecat labs
Eurecat Labs: more than 20 laboratories at the service of companies to find technological solutions to complex industrial or innovation challenges.

News
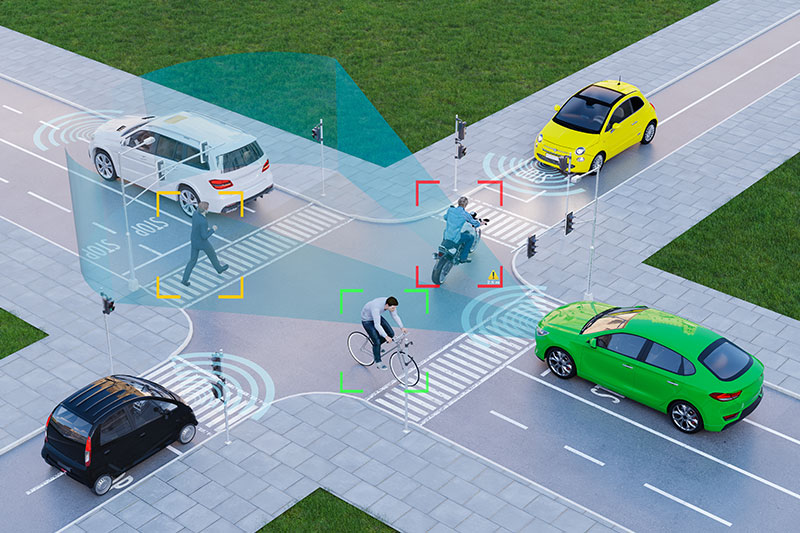
Cyber-resilience in cyber-physical systems: protecting the connected mobility of the future

The Eurecat technology centre expands its capabilities for the manufacture of more circular, recyclable and functional textiles

Cyber resilience in critical infrastructure: how to protect industrial systems against cyber-attacks

What are PFAS and how to remove them: technologies to reduce these “forever chemicals” in water

Emerging contaminants: a new challenge for water management

A smart orthodontic device that allows monitoring the time of use to facilitate treatment follow up
Activities agenda

04 JUN 2026
De 09:30 a 11:30
Diàleg LABIIT Repensant el turisme: fórmules per generar valor per al territori

08 JUN 2026
De 08:45 a 17:35
FORUM TURISTIC: el congrés sobre innovació i aplicació de les TIC al sector del turisme
El Forum TurisTIC és el congrés dedicat a la innovació i a l’aplicació de la tecnologia en el sector turístic, organitzat per Eurecat, Centre Tecnològic de Catalunya. El pròxim 8 de juny, a Barcelona, se celebrarà la catorzena edició sota el lema Innovació digital i turisme cultural: experiències transformadores i regeneratives. En aquesta edició, empreses del sector turístic i cultural, empreses d’innovació i desenvolupament [...]

09 JUN 2026
Del 09-06-2026 al 11-06-2026
DES 2026 Digital Enterprise Show
Eurecat és Partner del DES, principal esdeveniment internacional B2B sobre Intel·ligència Artificial i Tecnologies Exponencials per la transformació empresarial i estratègies de creixement de negocis. A Màlaga, entre els dies 9 i 11 de juny, els principals experts, empreses innovadores i líders del sector es reuneixen per compartir coneixements, explorar solucions disruptives i impulsar el [...]


16 JUN 2026
Del 16-06-2026 al 17-06-2026
5th Taulí Health – Artificial Intelligence Symposium THAIS 2026

16 JUN 2026
De 10:00 a 12:00
Workshop Presentación de los hubs sectoriales de la BSC AI Factory: impulso a la adopción de la IA en sectores estratégicos


01 OCT 2026
De 09:00 a 15:00
Congrés Discapacitat intel·lectual en l’era de la IA: La tecnologia com a motor d’una societat més inclusiva

27 OCT 2026
De 09:15 a 13:30









